|
With beryllium (Z 4), the 2s sublevel is complete and the 2p sublevel begins with. There are 4 valence electrons but only 2 electrons are unpaired. Draw the orbital filling diagram for carbon and write its electron. Also replace URL for the actual url of this page (The stay, ok?). If we consider carbon in the ground state, its electronic configuration will be 1s2 2s2 2p2. The portion of Carbon configuration that is equivalent to the. Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. The ground state abbreviated electronic configuration of Neutral Carbon atom is He 2s2 2p2. "Electron Configuration of Carbon (C) [Complete, Abbreviated, Uses. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: 
PDF Sorption of Carbon Dioxide at High Temperatures by Slag. The number and letter describe the energy level and orbital, and the number above the orbital. Which one of the following ions has electronic configuration. That gives credibility to your paper and it is sometimes required in higher education. For example, the electron configuration of lithium is 1s22s1. CitationWhen you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to maker your paper look cool?Īlong with basic atom / element information (like Carbon electron configuration and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Video Are you having trouble understanding the basics of atomic elements? This video will walk you through: Electron Configuration Oxidation States Plot Electronegativity (Pauling. After the 4s is full we put the remaining six electrons in the 3d orbital. For example, Carbon had an atomic weight of 12.00 in 1902 but today it is. In writing the electron configuration for carbon the first. History + Click the formula to re-roll a dice Settings + Quantity Colors Sound. Want to learn more details and data about Carbon (C)? Check my Elements Comprehensive List. A) Answer: 1s22s22p2 Explanation: Carbon is the sixth group element with a total of 6 electrons. Sixth most abundant element in the universe.ĭiscoveryDiscovered By: Known to the ancients Radiocarbon dating uses the carbon-14 isotope to date old objects.ĭescriptionAllotropic forms include diamonds and graphite. UsesFor making steel, in filters, and many more uses. SourcesMade by burning organic compounds with insufficient oxygen. 
In the case of Carbon the abbreviated electron configuration is 2s2 2p2. Nevertheless, check the complete configuration and other interesting facts about Carbon that most people don't know.Ĭomplete Electron Configuration1s2 2s2 2p2Ībbreviated Electron Configuration 2s2 2p2 Some are hard to memorise (or predict), so what is the electron configuration of an atom of C? You shouldīe able to identify both ground and excited state electron configurations.Įxample 1 : What is the ground state electronĪnswer : Carbon is the 6th element in the periodic table, hence the atomic number Z = 6, and therefore has 6 electrons.Electrons have a specific form of distribution (or configuration) in every atom, even Carbon. An electron configuration describes the arrangement. You should be familiar with how to determine an electronĬonfiguration for an atom and identify the valence electrons. Section 3 Electron Configurations Lesson Starter The electron configuration of carbon is 1s 2 2s 2 2p 2. 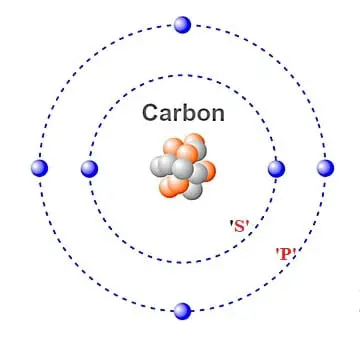
the electrons which travel furthest away from the nucleus) and the highestĬore electrons are electrons that under 'normal'Īre chemically inert and are in the completely filled inner energy levels. Full configuration, 1s2 2s2 2p Electron configuration chart. Valence electrons are the electrons utilised for bonding. Electron configuration for carbon Electron configuration. An excited state configuration is a higher energy arrangement (it requires energy input to create an excited state).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
